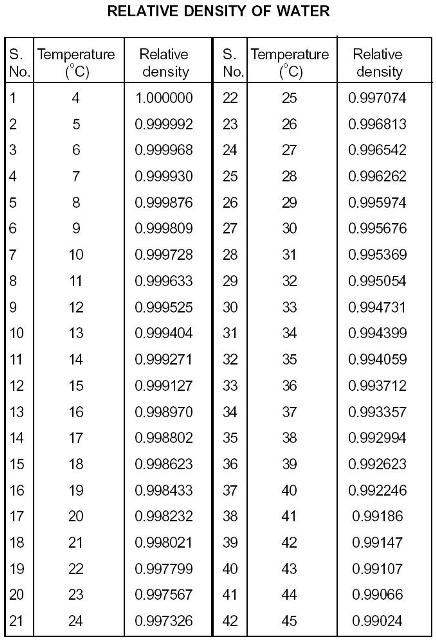
Due to this property, the density of ice is less than the density of water at 4 0 C (the maximum density). This behavior is called “density anomaly”. Above 4 0 C, the density of water decreases with increasing temperature. Unlike ordinary liquids, the density of water decreases from 4 0 C to 0 0 C. 
mass per unit volume of water has some interesting features, different from other liquids. One molecule of water (H 2 O) is composed of two hydrogen atoms and one oxygen atom, bound by two covalent bonds. Sulfur Dioxide Liquid - Thermal Propertiesĭensity, specific heat, thermal conductivity and more.Water is a tasteless, odorless transparent substance, which can be found in three states: liquid (water), solid (ice), and gaseous (vapor). The definition of STP - Standard Temperature and Pressure and NTP - Normal Temperature and Pressure. STP - Standard Temperature and Pressure and NTP - Normal Temperature and Pressure Specific heat of Nitrogen Gas - N2 - at temperatures ranging 175 - 6000 KĬhemical, Physical and Thermal Properties of Oxygen - O 2. Natural gas consumption for equipment like boiling pans, ovens, cookers, kettles and more. Phase diagram included.ĭefinition and molecular weight (molar mass) of some common substances. Specific heat of Hydrogen Gas - H2 - at temperatures ranging 175 - 6000 K.Ĭhemical, Physical and Thermal Properties of Hydrogen - H2.īoiling temperatures for common liquids and gases - acetone, butane, propane and more.Ĭhemical, Physical and Thermal Properties of Methane - CH 4. Specific gravities of air, ammonia, butadiene, carbon dioxide, carbon monoxide and some other common gases.Ĭhemical, Physical and Thermal Properties of Helium - He. Molar specific heats for some common gases at constant volume. Gases - Explosion and Flammability Concentration Limitsįlame and explosion limits for gases like propane, methane, butane, acetylene and more. 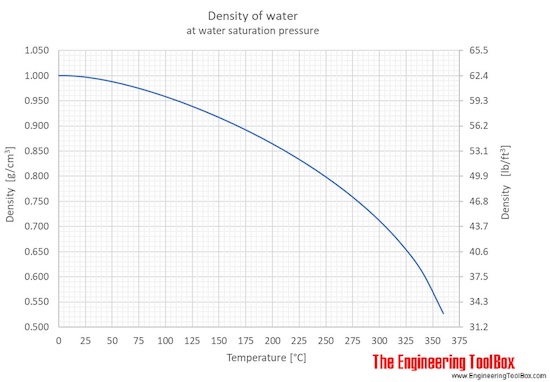
tolerable concentration levels for some industrial gases.Ībsolute (dynamic) viscosities of some common gases. 
Online density converter with commonly used units.ĭangerous vs. Potential biogas production from animal manure.Ĭhemical, physical and thermal properties of n-Butane.ĭensities of common products - Imperial and SI-units. Typical composition of biogas produced from household waste. Specific heat of Benzene Gas - C6H6 - at temperatures ranging 250 - 900 K.Ĭarbon - Nitrogen ratios for biogas produced from various raw materials. Material properties of gases, fluids and solids - densities, specific heats, viscosities and more.ĭry air is a mixture of gases where the average molecular weight (or molar mass) can be calculated by adding the weight of each component.īenzene Gas - Specific Heat vs. Definitions and convertion calculators.Īir, LNG, LPG and other common gas properties, pipeline capacities, sizing of relief valves. You can divide pounds per cubic foot by 32.2 for a rough value in slugs.ĭensities of solids, liquids and gases. Note that even if pounds per cubic foot is often used as a measure of density in the U.S., pounds are really a measure of force, not mass.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
